幼年二倍體和三倍體北極紅點鮭Salvelinus alpinus對窮盡運(yùn)動的生理反應(yīng)
The physiological response of juvenile diploid and triploid Arctic charr Salvelinus alpinus to exhaustive exercise
三倍體是生產(chǎn)不育魚的有效工具,但通常會導(dǎo)致商業(yè)水產(chǎn)養(yǎng)殖性能受損。有鑒于此,我們的研究比較了幼年二倍體和三倍體北極紅點鮭 Salvelinus alpinus 對窮盡運(yùn)動的生理反應(yīng),北極紅點鮭是一種具有巨大水產(chǎn)養(yǎng)殖潛力的極地物種。標(biāo)準(zhǔn)斜坡游泳方案顯示臨界游泳速度(U暴擊) 倍性之間。倍性對U后也沒有影響暴擊血糖、乳酸或血細(xì)胞比容。然而,三倍體的紅細(xì)胞核分割頻率明顯更高。與倍性無關(guān),血乳酸水平與U暴擊.我們得出的結(jié)論是,三倍體不會損害幼年高山沙門蟲對窮盡運(yùn)動的反應(yīng)。
水產(chǎn)養(yǎng)殖,紅細(xì)胞,運(yùn)動,極地,鮭科魚類,三倍體
Triploidy is an effective tool for producing sterile fishes but often results in impaired performance in commercial aquaculture. In light of this, our study compared the physiological response to exhaustive exercise in juvenile diploid and triploid Arctic charr Salvelinus alpinus, a polar species with great potential for aquaculture. A standard ramping swimming protocol revealed no significant difference in critical swimming velocity (Ucrit) between ploidies. There was also no effect of ploidy on post-Ucrit blood glucose, lactate or haematocrit. However, triploids had a significantly higher frequency of erythrocyte nuclear segmentation. Independent of ploidy, there was also a significant positive correlation between blood lactate levels and Ucrit. We conclude that triploidy does not impair the response to exhaustive exercise in juvenile S. alpinus.
aquaculture, erythrocyte, exercise, polar, salmonid, triploidy
1 引言
北極紅點鮭 Salvelinus alpinus L. 是一種冷水鮭魚,原產(chǎn)于北半球的北極、北方和溫帶地區(qū),由于其耐受高放養(yǎng)密度、低最佳飼養(yǎng)溫度以及高魚片質(zhì)量和產(chǎn)量,具有巨大的水產(chǎn)養(yǎng)殖潛力(S?ther 等人,2013 年;Yossa 等人,2019 年)。然而,養(yǎng)殖的高山葡萄容易在收獲前發(fā)生有性成熟,這一過程將能量分配從體細(xì)胞生長轉(zhuǎn)移到性腺,從而降低魚片產(chǎn)量和質(zhì)量,并增加疾病易感性(Yossa 等人,2019 年)。此外,正如養(yǎng)殖大西洋鮭魚 Salmo salar 所記錄的那樣,在發(fā)生養(yǎng)殖逃逸的情況下,它會產(chǎn)生馴化基因組基因滲入當(dāng)?shù)剡m應(yīng)野生種群的風(fēng)險(Glover 等人,2017 年;Wacker 等人,2023 年;Wringe 等人,2018 年)。
養(yǎng)殖魚類不必要的性成熟的實用解決方案是使用功能不育的三倍體種群(Benfey,2016 年;Piferrer 等人,2009 年)。然而,與二倍體相比,對三倍體鹽酸鏈球菌的廣泛中試和商業(yè)試驗已經(jīng)確定了許多福利和農(nóng)業(yè)生產(chǎn)問題,包括白內(nèi)障、骨骼畸形和總體生存率降低(Fraser 等人,2012 年;Madaro 等人,2022 年;O'Flynn 等人,1997 年)。盡管對三倍體高山鏈球菌的此類研究較少,但也有類似的證據(jù)表明存活率降低(Chiasson 等人,2009 年;Fraser 等人,2022 年)和椎體畸形發(fā)生率較高(Fraser 等人,2022 年)。許多研究還表明,三倍體鮭魚對高溫和缺氧等環(huán)境壓力的耐受能力降低(例如 Benfey 和 Devlin,2018 年;Hansen 等人,2015 年;Jensen 和 Benfey,2022 年;Sambraus 等人,2017 年、2018 年;Scott 等人,2015 年),可能是由于高溫下有氧范圍減少(Riseth 等人,2020 年)。然而,文獻(xiàn)中存在相互矛盾的發(fā)現(xiàn):至少有兩項研究未能發(fā)現(xiàn)三倍體對高溫下有氧范圍或最大代謝率的影響(Bowden 等人,2018 年;Sezaki 等人,1991 年)。
紅細(xì)胞大小的增加通常被認(rèn)為是三倍體呼吸能力的限制因素,因為與二倍體的較小紅細(xì)胞相比,這會導(dǎo)致相對于體積的表面積減少(Benfey,1999 年;Benfey 和 Devlin,2018 年)。三倍體鮭魚也經(jīng)常表現(xiàn)出較高的紅細(xì)胞核分割 (ENS) 發(fā)生率(Clark 等人,2025 年;Dorafshan 等人,2008 年;Wang 等人,2010 年;Wlasow 等人,2014 年,2004 年),一種來源不明的細(xì)胞畸形,定義為細(xì)胞核的內(nèi)側(cè)分裂(Yokote,1982)。 這種畸形尚未在 S. alpinus 中報道。
疫苗接種和分級等常規(guī)水產(chǎn)養(yǎng)殖作業(yè)要求魚類在短時間內(nèi)擁擠,通常與捕獲(通過網(wǎng))和從水中移走相結(jié)合。三倍體鮭魚在初級和次級應(yīng)激反應(yīng)方面通常對此反應(yīng)良好(Benfey & Biron,2000 年;Biron & Benfey,1994 年;Hyndman 等人,2003a;Madaro 等人,2024 年;Preston 等人,2017 年;Sadler 等人,2000 年),盡管如果在高溫下被推到筋疲力盡,三倍體可能會表現(xiàn)出高死亡率(Hyndman 等人,2003 年b)。
臨界游動速度 (U暴擊)是一種簡單的測試,經(jīng)常用于評估魚類對窮盡運(yùn)動的生理反應(yīng)。它與血細(xì)胞比容和血血紅蛋白水平(Pearson & Stevens,1991)、心輸出量(Clark & Seymour,2006)和代謝率(Horodysky 等人,2011 年;Norin &; Clark,2016 年),總體上反映了最大有氧能力(Kolok,1999 年; Norin 和 Clark,2016 年;Plaut,2001 年)。雖然之前的研究表明,U暴擊在幾種鮭魚物種中不受三倍體的影響(Bernier 等人,2004 年;Lijalad 和 Powell,2009 年;Riseth 等人,2020 年;Scott 等人,2015 年;Small &; Randall,1989 年;Stillwell &; Benfey, 1997),這尚未在 S. alpinus 中得到檢查。
在這項研究中,我們檢查了三倍體對這種極地物種對通過 U 進(jìn)行詳盡運(yùn)動的反應(yīng)的影響暴擊通過運(yùn)動后采血進(jìn)行測試。根據(jù)之前對其他鮭魚的研究,我們假設(shè) U暴擊二倍體和三倍體之間沒有區(qū)別。我們試圖通過測量繼發(fā)性應(yīng)激反應(yīng)(血糖、乳酸和血細(xì)胞比容)來補(bǔ)充這些信息,并調(diào)查 S. alpinus 中 ENS 的存在和潛在原因。這些信息可以幫助為這種相對未充分研究的鮭魚水產(chǎn)養(yǎng)殖物種制定特定物種的養(yǎng)殖場管理實踐。
2 材料與方法
2.1 道德聲明
這項研究得到了新不倫瑞克大學(xué) (UNB) 動物護(hù)理委員會(動物使用協(xié)議 24007)的批準(zhǔn),并遵循了加拿大動物護(hù)理委員會的所有適用福利和實驗指南。
2.2 魚
二倍體和三倍體 S. alpinus 胚胎是在眼卵階段從 Valorēs 育種計劃(加拿大 Shippagan,NB)購買的。它們是2023年4月人工產(chǎn)卵的3只母(池卵)和10只父(池白子)的后代,在受精后210°C-min開始,通過65.5 MPa的靜水壓力機(jī)處理5 min誘導(dǎo)了大約一半的卵,其余的保留為二倍體對照。三倍體的眼期存活率(發(fā)貨前)為 58 ± 1%,二倍體為 77 ± 1%。隨后,按照標(biāo)準(zhǔn)飼養(yǎng)程序(Jobling 等人,2010 年)在 UNB 水生設(shè)施(加拿大新罕布什爾州弗雷德里克頓)飼養(yǎng)魚類,二倍體和三倍體分別在供應(yīng)脫氯市政水的流通池中飼養(yǎng)。UNB 的死亡率沒有記錄,但很低,倍性之間沒有明顯差異。
2024 年 1 月,在實驗前的幾個月里,所有魚都被轉(zhuǎn)移到六缸循環(huán)水產(chǎn)養(yǎng)殖系統(tǒng) (RAS) 內(nèi)的 314 L 倍性特異性水箱中。該RAS的水質(zhì)參數(shù)保持在推薦范圍內(nèi)(總氨氮和亞硝酸鹽均為<1.0 mg/L,硝酸鹽為0-400 mg/L,堿度為50-300 mg/L,硬度為>100 mg/L,pH 值為 6.5-8.5;Timmons 等人,2018 年),但單日低堿度 (43 ppm) 除外,通過增加 RAS 的補(bǔ)充水立即得到糾正。在整個 RAS 中,包括直到實驗期結(jié)束,溫度和溶解氧的平均溫度和溶解氧平均分別為 10.3°C ± 1.0°C 和 93% ± 5% 的空氣飽和度。放養(yǎng)密度約為 109 kg m?3在實驗時,個體平均體重約為160克(表1)。用于該實驗的所有魚都來自單個二倍體和單個三倍體水箱。實驗前20天,室內(nèi)季節(jié)性調(diào)整的光周期從12:12增加到13:11 h(明:暗),以模擬初夏的季節(jié)趨勢,以維持在單獨(dú)的RAS內(nèi)的親魚,并且這種13:11狀態(tài)在整個實驗期間保持。
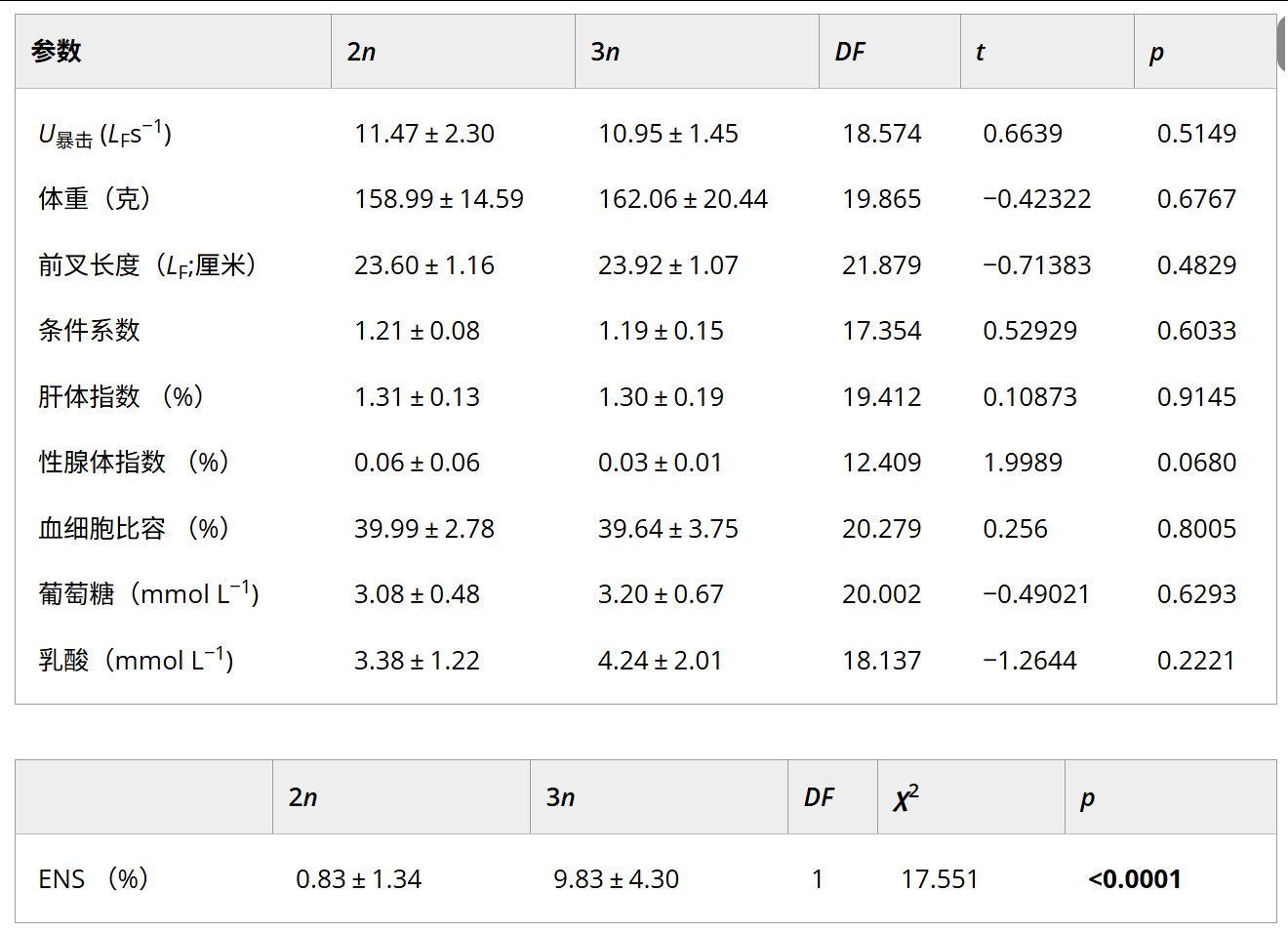
表 1. 臨界游動速度 (U暴擊)和魚到達(dá)U后立即測量的參數(shù)暴擊在幼年二倍體 (2n) 和三倍體 (3n) 中 Salvelinus alpinus(平均值 ± 標(biāo)準(zhǔn)差,每個倍性 n = 12)。
1 INTRODUCTION
The Arctic charr Salvelinus alpinus L. is a cold-water salmonid species native to Arctic, boreal and temperate regions of the northern hemisphere with great aquaculture potential due to its tolerance of high stocking densities, low optimum rearing temperature and high fillet quality and yield (S?ther et al., 2013; Yossa et al., 2019). However, farmed S. alpinus are prone to preharvest sexual maturation, a process which diverts energy allocation from somatic growth to the gonads, thereby decreasing fillet yield and quality, and increases disease susceptibility (Yossa et al., 2019). Additionally, it creates the risk of genetic introgression of the domesticated genome into locally adapted wild populations in the event of farm escapes, as documented for farmed Atlantic salmon Salmo salar (Glover et al., 2017; Wacker et al., 2023; Wringe et al., 2018).
A practical solution to unwanted sexual maturation of farmed fishes is to use functionally sterile triploid populations (Benfey, 2016; Piferrer et al., 2009). However, extensive pilot-scale and commercial trials with triploid S. salar have identified numerous welfare and farm production concerns when compared to diploids, including cataracts, skeletal deformities and overall reduced survival (Fraser et al., 2012; Madaro et al., 2022; O'Flynn et al., 1997). Although there have been fewer such studies of triploid S. alpinus, there is similar evidence of reduced survival (Chiasson et al., 2009; Fraser et al., 2022) and higher incidence of vertebral deformities (Fraser et al., 2022). Many studies have also shown triploid salmonids to have a reduced ability to tolerate environmental stressors such as high temperature and hypoxia (e.g. Benfey & Devlin, 2018; Hansen et al., 2015; Jensen & Benfey, 2022; Sambraus et al., 2017, 2018; Scott et al., 2015), possibly due to reduced aerobic scope at elevated temperatures (Riseth et al., 2020). There are, however, conflicting findings in the literature: at least two studies have failed to find such an effect of triploidy on aerobic scope or maximum metabolic rate at elevated temperatures (Bowden et al., 2018; Sezaki et al., 1991).
Increased erythrocyte size is often suggested as the limiting factor to the respiratory capacity of triploids because this results in reduced surface area relative to volume compared to the smaller erythrocytes of diploids (Benfey, 1999; Benfey & Devlin, 2018). Triploid salmonids also frequently exhibit a higher incidence of erythrocyte nuclear segmentation (ENS) (Clark et al., 2025; Dorafshan et al., 2008; Wang et al., 2010; Wlasow et al., 2014, 2004), a cellular deformity of unknown origin that is defined as the medial division of the nucleus (Yokote, 1982). This deformity has yet to be reported in S. alpinus.
Routine aquaculture operations such as vaccination and grading require fish to be crowded for short periods of time, often in combination with capture (by netting) and removal from the water. Triploid salmonids generally respond well to this in terms of their primary and secondary stress response (Benfey & Biron, 2000; Biron & Benfey, 1994; Hyndman et al., 2003a; Madaro et al., 2024; Preston et al., 2017; Sadler et al., 2000) although triploids may exhibit high mortality if pushed to exhaustion at high temperature (Hyndman et al., 2003b).
Critical swimming velocity (Ucrit) is a simple test frequently used to assess physiological responses to exhaustive exercise in fishes. It is positively correlated with haematocrit and blood haemoglobin levels (Pearson & Stevens, 1991), cardiac output (Clark & Seymour, 2006) and metabolic rate (Horodysky et al., 2011; Norin & Clark, 2016), and overall reflects maximum aerobic capacity (Kolok, 1999; Norin & Clark, 2016; Plaut, 2001). While previous studies have shown that Ucrit is not affected by triploidy in several salmonid species (Bernier et al., 2004; Lijalad & Powell, 2009; Riseth et al., 2020; Scott et al., 2015; Small & Randall, 1989; Stillwell & Benfey, 1997), this has not been examined in S. alpinus.
In this study, we examine the effects of triploidy on the response of this polar species to exhaustive exercise through a Ucrit test with post-exercise blood sampling. Based on previous studies in other salmonids, we hypothesized that Ucrit would not differ between diploids and triploids. We sought to supplement this information with measurements of secondary stress response (blood glucose, lactate and haematocrit), and to investigate the presence, and potential causes, of ENS in S. alpinus. Such information can assist in developing species-specific farm management practices for this relatively understudied salmonid aquaculture species.
2 MATERIALS AND METHODS
2.1 Ethics statement
This research was approved by the University of New Brunswick (UNB) Animal Care Committee (Animal Use Protocol 24007) and followed all applicable welfare and experimental guidelines of the Canadian Council on Animal Care.
2.2 Fish
Diploid and triploid S. alpinus embryos were purchased at the eyed-egg stage from the Valorēs breeding program (Shippagan, NB, Canada). They were progeny of three dams (pooled eggs) and 10 sires (pooled milt) artificially spawned in April 2023, with triploidy induced in approximately half of the eggs via hydrostatic pressure treatment of 5?min at 65.5?MPa beginning 210°C-min post-fertilization and the remainder retained as diploid controls. Survival to the eyed stage (prior to shipping) was 58?±?1% for triploids and 77?±?1% for diploids. Fish were subsequently reared at the UNB aquatic facility (Fredericton, NB, Canada) following standard husbandry procedures (Jobling et al., 2010), with diploids and triploids reared separately in flow-through tanks supplied with dechlorinated municipal water. Mortality at UNB was not recorded but was low and with no obvious difference between ploidies.
In January 2024, all fish were moved to 314-L ploidy-specific tanks within a six-tank recirculating aquaculture system (RAS) for the months preceding experimentation. Water quality parameters in this RAS were maintained within recommended ranges (total ammonia nitrogen and nitrite both <1.0?mg/L, nitrate 0–400?mg/L, alkalinity 50–300?mg/L, hardness >100?mg/L and pH?6.5–8.5; Timmons et al., 2018) with the exception of low alkalinity (43?ppm) on a single day that was immediately corrected by increasing makeup water to the RAS. Temperature and dissolved oxygen averaged 10.3°C?±?1.0°C and 93%?±?5% of air saturation, respectively, for the entire time in the RAS, including through to the end of the experimental period. Stocking density was approximately 109?kg?m?3 at the time of experimentation, with individuals weighing approximately 160?g on average (Table 1). All fish used for this experiment came from a single diploid and single triploid tank. Twenty days prior to experimentation, the seasonally adjusted photoperiod in the room was increased to 13:11?h (light:dark) from 12:12 to mimic the seasonal trends of early summer for the sake of broodstock maintained within a separate RAS, and this 13:11 regime was maintained throughout the experimental period.
TABLE 1. Critical swimming velocity (Ucrit) and parameters measured immediately after fish reached Ucrit in juvenile diploid (2n) and triploid (3n) Salvelinus alpinus (mean?±?standard deviation, n?=?12 per ploidy).
2.3 臨界游泳速度協(xié)議
試驗于 2024 年 6 月 5 日開始,連續(xù)持續(xù) 24 天。每天完成一次試驗,在三倍體和二倍體魚之間交替進(jìn)行,每個倍性總共 12 條魚。水下 30 升游泳隧道 (Loligo Systems) 用于確定 U暴擊,速度通過流量計校準(zhǔn)(Streamflo 430;尼克松流量計)。潛水泵與一個開放端口串聯(lián)使用,這兩個端口都位于電機(jī)驅(qū)動螺旋槳的正前方,以連續(xù)將淡水從外罐交換到游泳隧道中(10.7 L min?1).來自相鄰頭罐的恒定充氣水通過外罐泵送,以減輕游泳器材產(chǎn)生的任何熱量并確保穩(wěn)定的氧氣水平。溶解氧(Pro20;YSI)和溫度(可追溯 4015;Cole-Parmer)在外罐中測量。在 24 天的測試期間,游泳隧道裝置的平均溫度和溶解氧水平分別為 14.9°C ± 0.7°C 和空氣飽和度的 98% ± 2%。
將帶有反射表面的泡沫板夾在外池的外表面,以防止視覺刺激影響游泳行為。使用游泳室內(nèi)的自粘尺來估計每條魚的叉長(LE),從而計算游泳速度(LEs?1)以盡量減少實驗前的處理壓力。這個 LE高估了真正的前叉長度(LF;游泳試驗完成后測量)平均下降 3.9%。通過游泳池的透明蓋子從上方觀察魚,特別注意不要驚動魚。
游泳試驗每天在 0900 至 0920 之間開始,從其儲備池中隨意挑選當(dāng)天的魚,并放置在游泳隧道的游泳室(尺寸為 55 × 14 × 14 厘米)中。該種群中的剩余魚在取出測試魚后立即喂食,從而確保在選擇魚進(jìn)行實驗之前,每個種群中始終保留飼料 24 小時。一旦被放置在游泳隧道中,魚以 1.0 升的速度給予 1 小時的適應(yīng)期Es?1然后速度提高了1.0 LEs?1每 10 分鐘一次,遵循 Scott 等人(2015 年)的斜坡方案。這種情況一直持續(xù)到魚筋疲力盡,這被定義為無法逆流游泳并被迫在游泳池末端的金屬格柵上至少 3 秒。與 Scott 等人(2015 年)不同,我們沒有將魚作為 U 的一部分第二次游泳暴擊協(xié)議; 相反,記錄時間和游泳速度,關(guān)閉電機(jī),立即將魚從游泳隧道中取出并用苯佐卡因(4-氨基苯甲酸乙酯,0.05 g L)麻醉?1)在充氣的 3 L 浴中。電機(jī)關(guān)閉后,魚在腔室地板上保持一動不動,表現(xiàn)出疲憊的跡象,例如快速鰓蓋運(yùn)動,并且對處理反應(yīng)最小。24 條魚中的兩條(均為三倍體)在 U 處失去平衡暴擊;這些魚在移除和隨后的麻醉之前被允許在沒有流動的游泳隧道中恢復(fù)。
2.3 Critical swimming velocity protocol
Trials began on 5 June 2024 and continued for 24 consecutive days. One trial was completed per day, alternating between a triploid and diploid fish, for a total of 12 fish per ploidy. A submerged 30-L swim tunnel (Loligo Systems) was used to determine Ucrit, with velocity calibrated via a flow meter (Streamflo 430; Nixon Flowmeters). A submersible pump was used in tandem with an open port, both located immediately before the motor driven propeller, to continuously exchange fresh water from the outer tank into the swim tunnel (10.7?L?min?1). A constant flow of aerated water from an adjacent head tank was pumped through the outer tank to mitigate any heat generated by the swimming apparatus and to ensure stable oxygen levels. Dissolved oxygen (Pro20; YSI) and temperature (Traceable 4015; Cole-Parmer) were measured in the outer tank. Average temperature and dissolved oxygen levels in the swim tunnel setup over the 24-day test period were 14.9°C?±?0.7°C and 98%?±?2% of air saturation, respectively.
A foam board with a reflective surface was clamped to the exterior face of the outer tank to prevent visual stimuli from influencing swimming behaviour. A self-adhesive ruler within the swimming chamber was used to estimate each fish's fork length (LE) and thereby calculate swimming velocity (LEs?1) to minimize handling stress before beginning the experiment. This LE overestimated the true fork length (LF; measured after the swimming trial was completed) by an average of 3.9%. Fish were observed from above through the transparent lid of the swimming chamber, with special care taken not to startle the fish.
Swimming trials began between 0900 and 0920 every day with the fish of the day haphazardly chosen from its stock tank and placed in the swimming chamber (dimensions 55?×?14?×?14?cm) of the swim tunnel. The remaining fish in that stock tank were fed immediately after removing the test fish, thereby ensuring that feed was always withheld from each stock tank for 24?h prior to selecting a fish for experimentation. Once placed in the swim tunnel, the fish was given a 1-h habituation period at a velocity of 1.0?LEs?1 and then the velocity was increased by 1.0?LEs?1 every 10?min, following the ramping protocol of Scott et al. (2015). This continued until the fish was exhausted, which was defined as inability to swim against the current and being forced onto the metal grate at the end of the swimming chamber for at least 3?s. Unlike Scott et al. (2015), we did not swim the fish a second time as part of the Ucrit protocol; rather, the time and swimming velocity were recorded, the motor was switched off, and the fish was immediately removed from the swim tunnel and anaesthetized with benzocaine (ethyl-4-aminobenzoate, 0.05 g L?1) in an aerated 3-L bath. Fish remained motionless on the chamber floor after the motor was turned off, displaying signs of exhaustion such as rapid opercular movements and reacting minimally to handling. Two of the 24 fish (both triploids) lost equilibrium at Ucrit; these fish were allowed to recover in the swim tunnel with no flow before removal and subsequent anaesthesia.

