受精后不同時間靜水壓力沖擊誘導溪鱒魚三倍體血細胞的形態特征
Abstract
This study showed differences in the occurrence of blood cell alteration in triploid brook trout. The triploidisation was induced by hydrostatic shock pressure at 9500 psi for 5 min at different times after fertilisation (22.5, 27.5, 32.5, 37.5, 42.5, 47, 5, 52.5 and 62.5 min). The pressure shocks at 32.5 and 37.5 min after fertilisation caused the lowest share of pathologically altered blood cells in triploid fish.
這項研究表明,三倍體溪鱒魚血細胞改變的發生存在差異。受精后不同時間(22.5、27.5、32.5、37.5、42.5、47、5、52.5和62.5 min)在9500 psi下靜水沖擊壓力5 min誘導三倍化。受精后 32.5 分鐘和 37.5 分鐘的壓力沖擊導致三倍體魚中病理改變的血細胞比例最低。
關鍵字:
魚 基因組作 Salvelinus fontinalis 紅細胞 白細胞
Introduction
Triploidisation is a widely used method for producing sterile fish populations. The most effective method of production of triploid fish is application of pressure shocks (Pandian and Koteeswaran Citation1998). However triploid red tilapia was produced both by heat shocks and cold shocks (Pradeep, Srijaya, Bahuleyan et al. Citation2012; Pradeep, Srijaya, Papini et al. Citation2012). Selection of appropriate technical parameters for the experimental treatment of different fish species is not easy, and requires a lot of experience and skills to check the final result, for example ploidy verification in experimental groups of fish using cytogenetic or cytometric methods (Benfey et al. Citation1984; Johnstone and Lincoln Citation1986). In the red tilapia, identification of triploid individuals was conducted based on nuclear volume, cytoplasmic volume and nucleus surface area of erythrocytes (Pradeep et al. Citation2011). The most suitable method for ploidy verification in brook trout, Salvelinus fontinalis, seems to be an erythrocyte dimensions study from blood smears (Woznicki and Kuzminski Citation2002).
In triploid fish, produced by pressure or thermal shock, the alterations in erythrocytes, haemoglobin and red cell indices were observed (Benfey and Sutterlin Citation1984b; Benfey et al. Citation1984; Benfey Citation1999; Strunjak-Perovic et al. Citation2003; Wlasow et al. Citation2004; Dorafshan et al. Citation2008; Wang et al. Citation2010; Wlasow and Fopp-Bayat Citation2011). Triploidisation also influenced the changes in white blood cells, immunological defence reactions and stress response (Svobodová et al. Citation1998, Citation2001 Benfey and Biron Citation2000; Beyea et al. Citation2005; Maxime Citation2008; Fraser et al. Citation2012).
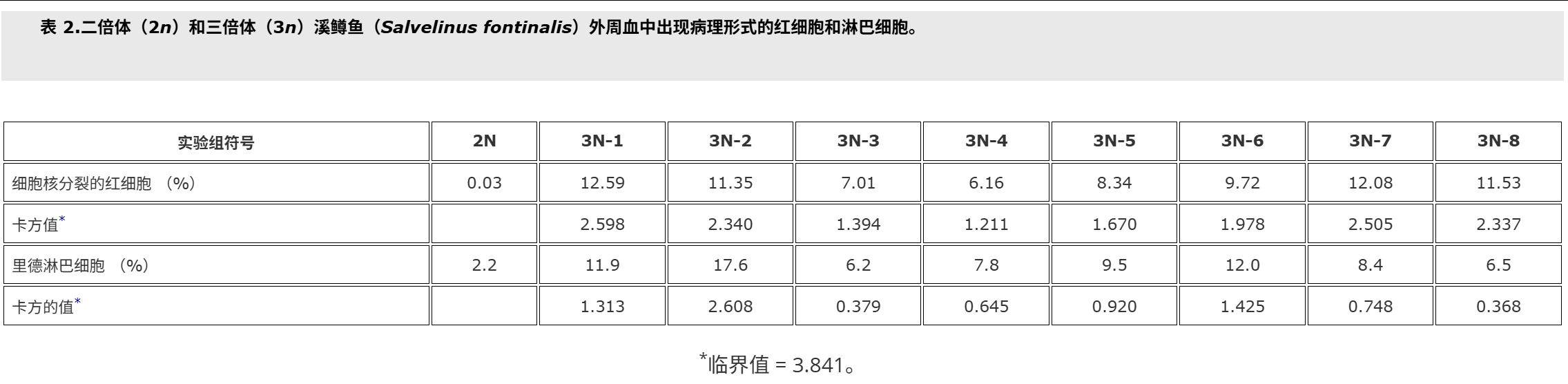
In the peripheral blood of triploid brook trout the percentage of red blood cells with divided nuclei were statistically significantly higher compared with the control group (19.1% versus 0.3%) (Wlasow et al. Citation2004). However, in this study the effect of triploidisation caused by shocks applied at 20 minutes after fertilisation was shown, but the influence of other technical parameters was not examined. Therefore the first purpose of the present study was to investigate the effect of differential time from fertilisation to pressure shock on the blood cell alteration in triploid brook trout. The second aim of the study was to estimate the minimal number of erythrocytes necessary for determination of percentage of red cells with divided nuclei in peripheral blood of brook trout triploids.
三倍化是一種廣泛使用的生產不育魚種群的方法。生產三倍體魚最有效的方法是施加壓力沖擊(Pandian 和 Koteeswaran引文1998).然而,三倍體紅羅非魚是由熱沖擊和冷沖擊產生的(Pradeep、Srijaya、Bahuleyan 等人。引文2012;普拉迪普、斯里賈亞、帕皮尼等人。引文2012).為不同魚類的實驗處理選擇合適的技術參數并不容易,需要大量的經驗和技能來檢查最終結果,例如使用細胞遺傳學或細胞術方法對魚類實驗組進行倍性驗證(Benfey 等人。引文1984;約翰斯通和林肯引文1986).在紅羅非魚中,根據紅細胞的核體積、細胞質體積和細胞核表面積進行三倍體個體的鑒定(Pradeep 等人。引文2011).最合適的溪鱒魚倍性驗證方法,Salvelinus fontinalis,似乎是通過血涂片進行紅細胞尺寸研究(Woznicki 和 Kuzminski引文2002).
在由壓力或熱沖擊產生的三倍體魚中,觀察到紅細胞、血紅蛋白和紅細胞指數的改變(Benfey 和 Sutterlin引文1984年b;本菲等人。引文1984;本菲引文1999;Strunjak-Perovic 等人。引文2003;Wlasow 等人。引文2004;多拉夫山等人。引文2008;王等人。引文2010;Wlasow 和 Fopp-Bayat引文2011).三倍化還影響白細胞、免疫防御反應和應激反應的變化(Svobodová 等人。引文1998,引文2001本菲和比隆引文2000;貝亞等人。引文2005;馬克西姆引文2008;弗雷澤等人。引文2012).
在三倍體溪鱒魚的外周血中,與對照組相比,細胞核分裂的紅細胞百分比在統計學上顯著更高(19.1% 對 0.3%)(Wlasow 等人。引文2004).然而,在這項研究中,顯示了受精后 20 分鐘施加的沖擊引起的三倍化的影響,但沒有檢查其他技術參數的影響。因此,本研究的首要目的是研究從受精到壓力休克的時間差異對三倍體溪鱒魚血細胞改變的影響。該研究的第二個目的是估計測定溪鱒魚三倍體外周血中細胞核分裂的紅細胞百分比所需的最小紅細胞數量。
材料和方法
Material and methods
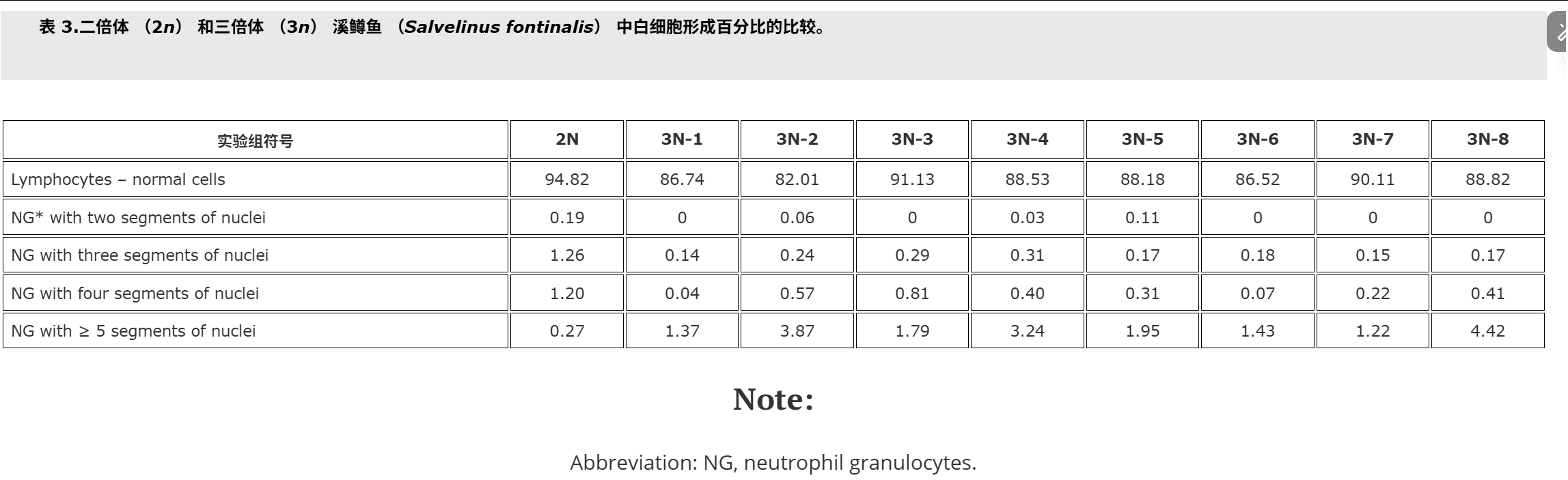
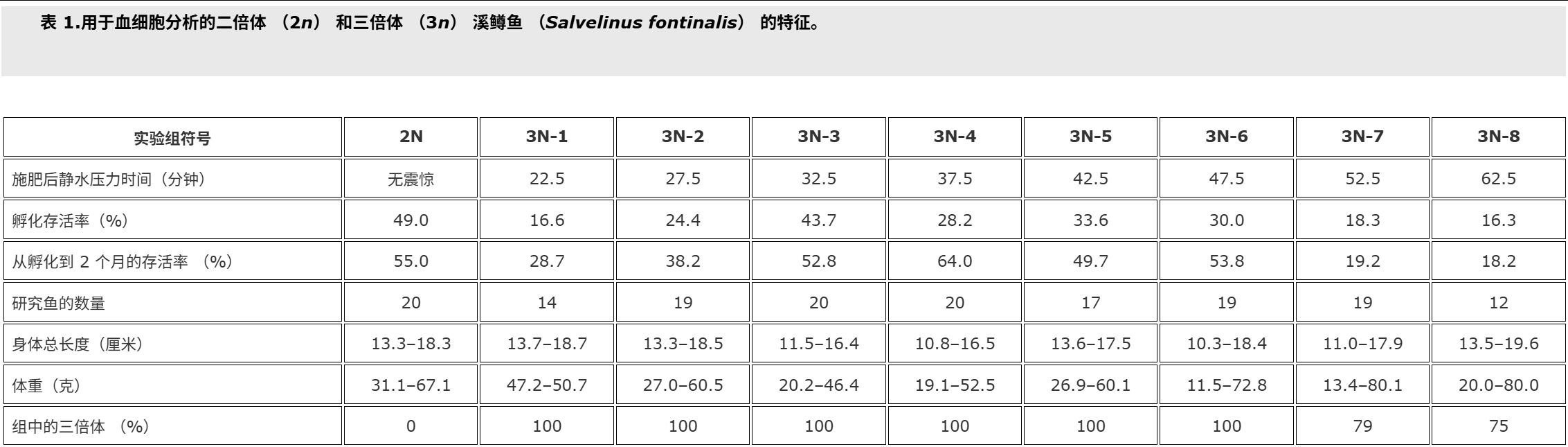
Fish used for this study were cultured at the Salmonid Research Department in Rutki (Institute of Inland Fisheries in Olsztyn, Poland). Both diploids (2n) and triploids (3n) came from the same lot of eggs. Triploid brook trout Salvelinus fontinalis (Mitchill) were obtained by using hydrostatic pressure shock of 9500 psi (65.5 × 103 kPa) on eggs at 10°C (Deeley and Benfey Citation1995). The pressure shock duration was 5 minutes, but time from fertilisation to shock varied in the experimental groups, and ranged from 22.5 min (group 3N-1) to 62.5 min. (group 3N-8, Table ).The diploid (control – 2N, 3N) received no pressure shock. Fish of the experimental groups were kept in separate tanks supplied with river water. During the experiment prophylactic chloramine baths were used twice a week. These baths are carried out during each rearing of fish at the Salmonid Research Department in Rutki (Institute of Inland Fisheries in Olsztyn, Poland) due to the presence of pathogens in the water supply hatchery and rearing system. Fish were fed granulate feed BioMar (Denmark). Survival from hatching to the collection of blood is shown in Table . Two months after hatching blood was collected from the caudal vessels to heparinised syringes. Propiscin (0.2% etomidate, Inland Fisheries Institute in Olsztyn, Poland) was used as an anaesthetic (0.5 ml l?1 of water for 10 min) in all experimental groups. Fish expected to be triploids (eight experimental groups, Table ) and diploids (20 specimens) were used for preparing blood smears. Smears were fixed in 95% methanol for 3 minutes, left to air dry and stained with 20% Giemsa solution for 15 minutes. The percentage of erythrocytes with divided nuclei was determined based on the observation of 300 cells from two slides for fish. For the study of white blood cells 200 cells were analysed, and the following forms have been studied: normal lymphocytes, Rieder’s lymphocytes (cells with divided nuclei), and neutrophil granulocytes (NGs) with 2, 3, 4 and ≥?5 segments of nuclei. The identification was based on the leukocyte descriptions published by Lehmann and Stürenberg (Citation1975).
用于本研究的魚是在魯特基的鮭魚研究部(波蘭奧爾什丁內陸漁業研究所)養殖的。二倍體 (2n) 和三倍體 (3n) 都來自同一批卵。三倍體溪鱒魚 Salvelinus fontinalis (Mitchill) 是通過在 10°C 下對雞蛋進行 9500 psi (65.5 × 103 kPa) 的(上海瑾瑜出售)靜水壓力機沖擊獲得的 (Deeley 和 Benfey)引文1995).壓力休克持續時間為5 min,但實驗組從受精到休克的時間各不相同,從22.5 min(3N-1組)到62.5 min不等(3N-8組,表).二倍體(對照 – 2N、3N)沒有受到壓力沖擊。實驗組的魚被飼養在單獨的水箱中,供水。在實驗期間,每周使用兩次預防性氯胺浴。由于供水孵化場和飼養系統中存在病原體,這些浴是在 Rutki 的鮭魚研究部(波蘭奧爾什丁內陸漁業研究所)每次養魚期間進行的。魚被喂食顆粒飼料 BioMar(丹麥)。從孵化到采血的存活率見表.孵化后兩個月,從尾部血管收集血液到肝素注射器中。使用普羅匹辛(0.2% 依托咪酯,波蘭奧爾什丁內陸漁業研究所)作為麻醉劑(0.5 毫升升?1水 10 分鐘)在所有實驗組中。預計為三倍體的魚(八個實驗組,表)和二倍體(20個標本)用于制備血涂片。將涂片固定在95%甲醇中3分鐘,風干并用20%吉姆薩溶液染色15分鐘。根據對兩張魚類載玻片的 300 個細胞的觀察,確定具有分裂細胞核的紅細胞百分比。為了研究白細胞,分析了 200 個細胞,并研究了以下形式:正常淋巴細胞、里德淋巴細胞(細胞核分裂的細胞)和具有 2、3、4 和 ≥ 5 段細胞核的中性粒細胞 (NG)。鑒定基于 Lehmann 和 Stürenberg 發表的白細胞描述(引文1975).
Simultaneously chromosome preparations (10 specimens from each experimental group) and the erythrocyte nuclei major axis measurements (all fish from each treatment) were done using procedure described by Woznicki and Kuzminski (Citation2002) in order to confirm fish ploidy. Differential cell counts were made under a 1000 × magnification.
Statistical analyses were performed on data from microscopic observations as the actual number of segmented nuclei in analysed preparations. In Tables and data were summarised in the form of percentages. All statistical analyses were evaluated at a significance level of 0.05. The chi-square test was used to evaluate the differences in proportion of “abnormal” to normal erythrocytes and leukocytes in triploid and diploid trout. Statistical differences between the observed number of red blood cells with segmented nucleus in the groups of 2N and 3N fish were calculated based on an alternative non-parametric analysis of variance – ANOVA Kruskal–Wallis (α = 0.05). In order to investigate how many nuclei should be counted to show the dependence of the number of nuclei divided by ploidy the Friedman ANOVA test was used. Data were collected from 71 fish, step by step as follows: 50 nuclei were observed and selected as normal and divided, and then the next 50 nuclei were observed and also selected as normal and divided (two or more parts), after that the cyclic analysis of next 50 nuclei was conducted. A total of 300 nuclei were counted. It was hypothesised that the results obtained for the count of 50, 100, 150, 200, 250 and 300 nuclei did not differ significantly and therefore the study of ploidy is sufficient to perform only counts of 50 nuclei.
同時使用 Woznicki 和 Kuzminski 描述的程序進行染色體制備(每個實驗組 10 個標本)和紅細胞核長軸測量(來自每個處理的所有魚)(引文2002)以確認魚的倍性。在 1000 ×放大倍率下進行差異細胞計數。
對來自顯微觀察的數據進行統計分析,作為分析制劑中分段細胞核的實際數量。在表格中和數據以百分比的形式匯總。所有統計分析均以 0.05 的顯著性水平進行評估。采用卡方檢驗評估三倍體和二倍體鱒魚中“異常”與正常紅細胞和白細胞比例的差異。根據替代非參數方差分析 - 方差分析 Kruskal-Wallis (α = 0.05) 計算 2N 和 3N 魚組中觀察到的具有分段細胞核的紅細胞數量之間的統計差異。為了研究應該計算多少個細胞核以顯示細胞核數除以倍性的依賴性,使用了弗里德曼方差分析檢驗。從71條魚中收集數據,逐步如下:觀察50個細胞核,選擇正常并分裂,然后觀察接下來的50個細胞核,也選擇為正常并分裂(兩個或多個部分),然后對接下來的50個細胞核進行循環分析。總共計算了 300 個細胞核。據推測,對 50、100、150、200、250 和 300 個細胞核的計數獲得的結果沒有顯著差異,因此倍性研究足以僅對 50 個細胞核進行計數。